A Field Trial Evaluating the Health and Performance of Lambs Fed a Bovine Colostrum Replacement
Berge AC, Hassid G, Leibovich H, Solomon D, Haines DM, Chamorro MF
DOIDOI: 10.21767/2572-5459.100044
Berge AC1*, Hassid G2, Leibovich H3, Solomon D4, Haines DM5 and Chamorro MF6
1Department of Reproduction, Obstetrics and Population Health, Faculty of Veterinary Medicine, Ghent University, Merelbeke, Belgium
2S.H.Design Engineering Ltd, Herzliya, Israel
3Extension Small Ruminant Production Systems, Moshav Udim, Israel
4Hachaklait Veterinary Services, Israel
5Department of Veterinary Microbiology, Western College of Veterinary Medicine, University of Saskatchewan, Saskatoon, Canada
6Department of Clinical Sciences, College of Veterinary Medicine, Kansas State University, Kansas, USA
- *Corresponding Author:
- Berge AC
Department of Reproduction, Obstetrics and Population Health
Faculty of Veterinary Medicine
Ghent University; Merelbeke, Belgium
Tel: +32499703112
E-mail: anna.berge@ugent.be
Received date: March 30, 2018; Accepted date: April 13, 2018; Published date: April 20, 2018
Citation: Berge AC, Hassid G, Leibovich H, Solomon D, Haines DM, et al. (2018) A Field Trial Evaluating the Health and Performance of Lambs Fed a Bovine Colostrum Replacement. J Anim Res Nutr Vol No 3: Iss no: 1: 6.
Copyright: © 2018 Berge AC, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Context: Failure of passive transfer of immunity due to insufficient and/or delayed administration of good quality colostrum is a risk for disease, death and reduced weight gain in lambs.
Objective: This trial evaluated the performance and health of lambs fed a bovine source lacteal derived colostrum powder in addition to farm colostrum compared to two feedings of farm colostrum.
Setting: This trial was performed on a large commercial sheep farm in Israel in 2015.
Participants: Two cohorts, a total of 60 male lambs, were enrolled at birth and followed until sent to market at approximate 5.5 months of age.
Interventions: The lambs randomly assigned at birth to receive either 250 ml of a bovine colostrum replacement (CR) or 250 ml of farm source ewe colostrum (FC) within half an hour of birth. Within 6 hours of birth a second feeding of 250 ml of farm source ewe colostrum was administered to both groups.
Main outcome measures: Colostral Brix% and serum brix% was measured using a Brix digital refractometer and compared between treatments. Morbidity, mortality and weight gain was recorded until marketing at average 165 days of age and compared between treatments.
Results: The Brix values were significantly higher in CR compared to FC. Serum% Brix was significantly higher in CR than FC lambs. There was significantly less disease in CR lambs (0%) than in FC lambs (17%) (P=0.05). Significantly more CR lambs (97%, 29 of 30 lambs) were marketed compared to FC lambs (77%, 23 of 30 lambs) (P=0.05). Conclusions: The use of this commercial colostrum replacement may facilitate colostrum management and improve health and productivity in lambs.
Keywords
Lambs; Colostrum; Passive transfer of immunity; Health; Growth rate; Mortality
Introduction
High neonatal morbidity and mortality remains a worldwide challenge that impacts lamb welfare and farm productivity [1]. Adequate colostrum intake is a key to reducing losses due to infectious disease in newborn lambs [2]. However, failure in the transfer of passive immunity remains the major cause of morbidity and mortality in small ruminants. Successful and efficient rearing systems wherein lambs are separated from their dams at birth and fed calf milk replacer for production of marketable sheep milk have been reported [3,4]. A commercial bovine source colostrum replacement product could be used on sheep farms to replace or supplement maternal colostrum. There are studies showing that the efficiency of absorption of IgG from bovine colostrum is similar to ovine colostrum [5]. However, field studies evaluating the health and growth outcomes in lambs receiving bovine colostrum as a replacement or supplement for maternal colostrum are lacking. The aim of this clinical field trial was to evaluate the performance and health of lambs given a first feed of a bovine origin lactealderived colostrum powder compared lambs fed similar volumes of farm source ewe colostrum on morbidity, mortality, weight gain and marketing of lambs.
Material and Methods
Study site
This trial was conducted in the Lachish region in Israel, at a commercial sheep farm with 700 dams. All lambs were separated from the ewe immediately after birth and hand fed colostrum, and thereafter ad lib bovine milk replacer (23% protein and 20% fat), through an automatic milk feeding system in indoor group housing units. During the preweaning period the lambs were also offered fresh vetch hay and starter pellets. After weaning, lambs were fed 22% crude proteins pellets and whole barley grains at a 1:1 ratio ad lib. Lambs were sent to market at approximately 5.5 months of age.
Study cohorts
Two cohorts of male lambs were enrolled in the study, the first cohort was enrolled from August 22nd-30th in 2015, and the second cohort from November 2nd-7th in 2015. The lambs were separated from the dam immediately after birth, ear tagged and weighed. Lambs weighing 1.5 Kg or more with no signs of physical abnormalities were enrolled in trial.
Treatment
Newborn lambs were randomly assigned to either the CR or FR treatment to receive either 250 ml of bovine origin lactealderived colostrum replacement (CR) from Saskatoon Colostrum Company Ltd (SCCL) or 250 ml of thawed and warmed previously frozen pooled farm ewe’s colostrum (FC). The CR was constituted using 75 g SCCL colostrum powder mixed into 200 ml water at 40°C and the farm colostrum was thawed and fed at similar temperature. The first colostrum feeding was within 30 minutes of birth and was performed by the farm’s veterinarian or the veterinary assistant. Two to four hours after the first colostrum feeding, all lambs received an additional feeding of 250 ml of FC for a total of 500 ml of colostrum fed within 6 hours of birth. After the two colostrum feeds the lambs were transferred to a group pen where they received the same milk replacer and grain supplement ration until weaning and marketing.
Samplings and study outcomes
Colostrum samples (first and second feeding) were collected. Blood samples were collected in serum tubes from all lambs 24 hours after birth. Serum samples and colostrum samples were tested for total solids using a digital Brix refractometer. Lambs were weighed at birth prior to colostrum ingestion, at weaning (at approx. 4 weeks of age), and on the day sent to market at approximately 165 days of age. The exact date of weighing was recorded and the weight at birth was recorded to the nearest 0.1 Kg. Weight gain performance was measured as average daily gain (ADG). Lamb health was evaluated daily by the farmer who was blinded to treatment allocation, and confirmation of diagnosis and treatment of sick lambs were made by veterinarian and veterinary assistant. All health events preweaning were recorded including clinical signs, treatments administered and mortality. After weaning, the farmer disposed of lambs with poor performance (slaughter, sell or give away). After weaning, mortality events could have been unreported as poor doing animals had been removed and therefore the lambs marketed was used as the final performance outcome.
Statistical Analysis
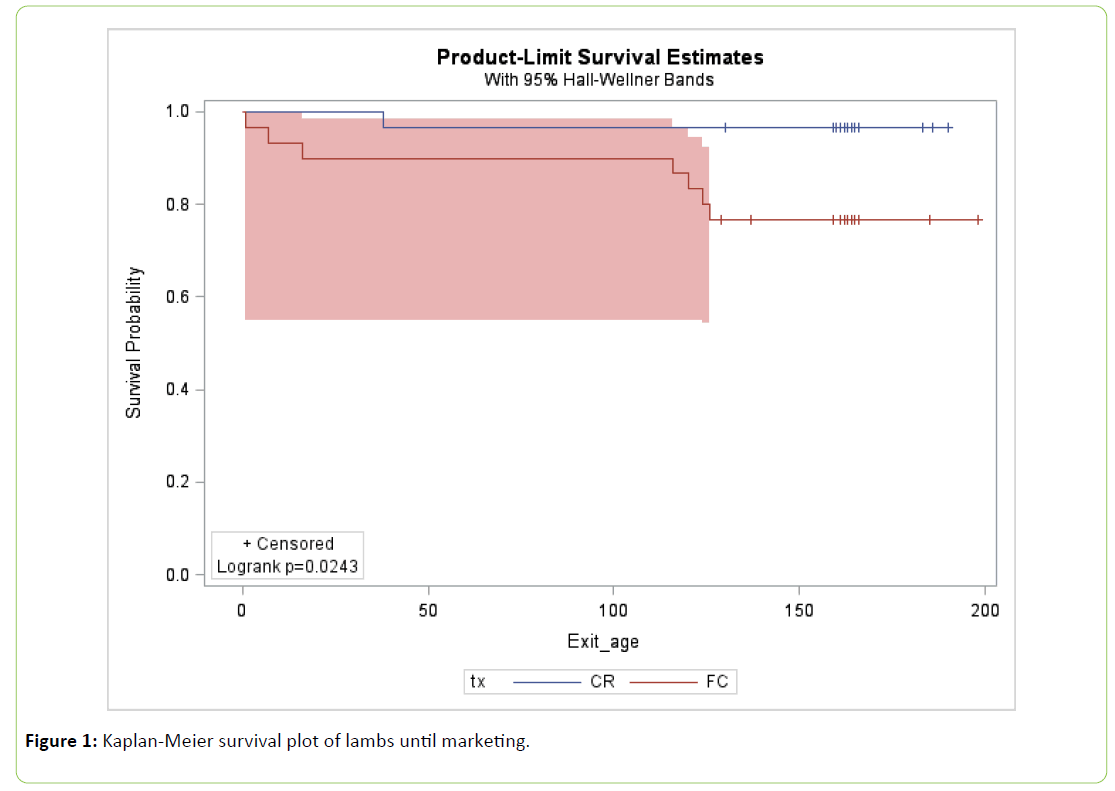
Study data were entered into an excel spreadsheet, verified and summary statistics and statistical analysis was done with SAS version 9.4. The outcomes evaluated were; morbidity, mortality, serum-Brix, average daily gain as measured from birth to weaning (mean 34 days), at 4 months (mean 127 days) and at marketing (mean 165 days). General linear models (GLM) including cohort as covariates were used to evaluate colostral brix %, serum brix % and birth weight. GLM including cohort and birth weight and age of lamb as covariates were used to evaluate ADG from birth to 4 months and ADG from birth to marketing. Least square means (LSM) and standard errors (SE) from the GLM are presented. The incidences of disease and mortality and the number of lambs that were marketed were compared by a stratified table and the difference in disease incidence evaluated using Fisher's exact test. Kaplan-Meier Plots were used to describe the lambs that were removed from the study or died prior to marketing.
Results
The first cohort consisted of with 11 CR lambs and 11 FC lambs. The second cohort contained 19 CR lambs and 19 FC lambs. For the first feed the %Brix of the CR (26.4 ± 0.2) was significantly higher (P<0.01) than the %Brix of FC (15.7 ± 0.2) fed to as first feed (Table 1). For the second feed farm colostrum the % Brix fed to CR lambs (15.5 ± 0.1) was not significantly different to FC lambs (15.7 ± 0.1) (P=0.55). The %Brix in serum of CR lambs (8.6 ± 0.1) was significantly higher than the %Brix of the serum of FC lambs (7.8 ± 0.8) (P<0.01) (Table 1).
| Colostrum brix % | Lamb serum | |||
|---|---|---|---|---|
| Treatment | Cohort | Feed 1 | Feed 2 | Brix % |
| CR | 1 | 26.8 | 16.8 | 8.5 |
| CR | 2 | 25.8 | 14.2 | 8.6 |
| FC | 1 | 17.1 | 16.9 | 8 |
| FC | 2 | 14.5 | 14.4 | 7.6 |
Table 1: Summary of colostrum and serum Brix
In Cohort 1,4 FC lambs were diagnosed with respiratory disease and two of these lambs died, whereas no lambs in the CR group were diagnosed with disease or died during the study period (Table 2). In cohort 2, two FC lambs showed depression in the first week of life and one of these lambs died, whereas no CR lambs showed signs of disease or died. There was significantly less disease in CR lambs (0%) compared to FC lambs (17%) (P=0.05) (Table 2). The preweaning mortality in CR lambs (0%) was not significantly lower than FC lambs (7%) (P=0.24). Twenty-three of the 30 FC lambs (23%) were marketed, whereas 29 of the 30 CR lambs (3%) was marketed (P=0.05) (Table 2).
| Preweaning Disease | Preweaning Dead | Lambs marketed | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Treatment | No | Yes | % | No | Yes | % | Yes | No | % |
| CR | 30 | 0 | 0.00% | 30 | 0 | 0.00% | 29 | 1 | 96.70% |
| FC | 25 | 5 | 16.70% | 28 | 2 | 6.70% | 23 | 7 | 76.70% |
| P = 0.05 | P = 0.24 | P = 0.05 | |||||||
Table 2: Pre-weaning morbidity and mortality and lambs sent to market.
Survival analysis of removal or death from the flock prior to marketing confirmed that significantly more FC lambs left the group than the CR lambs (P=0.02) (Figure 1). The ADG of CR lambs at weaning was 0.355 ± 0.013 Kg/d and not significantly higher than the ADG of FC lambs 0.332 ± 0.014 Kg/d (P=0.22) (Table 3). Similarly, at marketing the ADG of CR lambs was 0.428 ± 0.010 Kg/d and not significantly higher than the ADG of FC lambs 0.413 ± 0.011 Kg/d (P=0.29) (Table 3).
| Birth Weight (Kg) | Weaning ADG (Kg) | Marketing ADG (Kg) | |||||
| Treatment | Cohort | Mean | Std | Mean | Std | Mean | Std |
| CR | 1 | 4.145 | 1.06 | 0.335 | 0.06 | 0.414 | 0.036 |
| CR | 2 | 3.932 | 0.96 | 0.374 | 0.083 | 0.446 | 0.042 |
| FC | 1 | 4.127 | 1.292 | 0.332 | 0.107 | 0.386 | 0.113 |
| FC | 2 | 3.821 | 1.241 | 0.339 | 0.084 | 0.441 | 0.045 |
Table 3: Summary of Birth weight, and average daily gain at weaning and at marketing.
Discussion
This study indicates that feeding newborn lambs a bovine derived colostrum replacement product to replace the first ewe colostrum feeding resulted in higher serum %Brix, decreased morbidity, a trend for decreased preweaning mortality and an overall increase in the number of lambs marketed.
The ewe colostrum used in this study had relatively low %Brix likely indicating lower levels of immunoglobulin relative to the bovine CR used to supplement the lambs in the CR group. In this study, the CR lambs had higher serum Brix% compared to FC lambs. There are numerous studies indicating that there is a high correlation between %Brix and serum IgG in dairy calves [6,7]. For the purpose of this study, the serum Brix values gave some indication to levels of IgG in the serum of the lambs. Furthermore, this Brix measure does not present differential results based upon antibody species, and may thereby be indicative of total IgG, whether bovine or ovine source. Further research with IgG determination in colostrum and lamb serum is recommended.
Bovine colostrum has also been used to supplement goat kids and resulted in higher serum immunoglobulin status and improved growth [8]. Furthermore, bovine colostrum products may also be an alternative to ewe or doe colostrum to prevent transmission of maternal colostrum borne pathogens such as Maedi-Visna Virus and Scrapie to lambs or kids [9,10].
Early access to sufficient colostrum is crucial for neonatal lamb health [2]. In this study the lambs were fed a total of 500 mLs of colostrum divided into 2 feedings within the first 6 hours of life. The lambs that received the first feeding of 250 mLs of the bovine CR supplement (100 g IgG/470 g powder) received 16 g IgG in that feeding. Since the mean birth weight of the lambs was approximately 4 kg this is about 4 g IgG/kg delivered in the first feeding. In farms where the lambs are removed from the dam, it has been noted that feeding colostrum equivalent to 8 g of IgG/kg of body weight results in immunity comparable to lambs reared under natural conditions [11]. Other authors recommend total colostrum feeding of 30 g IgG per lamb [12]. In a study where 250 ml pooled bovine colostrum was fed in 2 meals compared to equal quantities of ovine colostrum, they found that the IgG content in the pooled bovine colostrum was more than twice as high as the ovine colostrum, and the study also indicated that the efficiency of absorption of colostral IgG was similar between bovine and ovine colostrum [5].
It has previously been noted significantly higher growth in kids that had been supplemented with bovine colostrum [8]. In this study, the ADG was not significantly different between CR and FC lambs. This could be partly a censoring bias due to that the farm removed (personal sales, gifts, slaughter) lambs that were not performing well and sick lambs with lower weight gains died in the FC group.
The use of this commercial colostrum replacement may facilitate colostrum management and improve health and productivity in lambs. Results from this study indicate that a commercial bovine dried colostrum product can be a valuable alternative or supplement to ewe colostrum to optimize health and performance in lambs reared artificially on milk replacers. Further studies are recommended to determine the optimal means to use dried bovine colostrum as means to improve productivity in lambs.
Acknowledgements
We would like to extend our gratitude to Haim Hirsch, owner of Hirsch farm in Israel. This study was financially supported by SH Design Engineering Ltd. Saskatoon Colostrum Company Ltd (SCCL) provided product support for this study.
Conflict of Interest Statement
Deborah Haines is Director of Research at SCCL.
References
- Dwyer CM, Conington J, Corbiere F, Holmøy IH, Muri K, et al. (2016) Invited review: Improving neonatal survival in small ruminants: Science into practice. Animal 10: 449-459.
- Nowak R, Poindron P (2006) From birth to colostrum: Early steps leading to lamb survival. Reprod Nutr Dev 46: 431-446.
- Bimczok D, Röhl FW, Ganter M (2005) Evaluation of lamb performance and costs in motherless rearing of German Grey Heath sheep under field conditions using automatic feeding systems. Small Rumin Res 60: 255-265.
- Emsen E, Yaprak M, Bilgin OC, Ockerman HW (2004) Growth performance of Awassi lambs fed calf milk replacer. Small Rumin Res 53: 99-102.
- Moretti DB, Kindlein L, Pauletti P, Machado-Neto R (2010) IgG absorption by Santa Ines lambs fed Holstein bovine colostrum or Santa Ines ovine colostrum. Animal 4: 933-937.
- Hernandez D, Nydam DV, Godden SM, Bristol LS, Kryzer A, et al. (2016) Brix refractometry in serum as a measure of failure of passive transfer compared to measured immunoglobulin G and total protein by refractometry in serum from dairy calves. Vet J 211: 82-87.
- Morrill KM, Polo J, Lago A, Campbell J, Quigley J, et al. (2013) Estimate of serum immunoglobulin G concentration using refractometry with or without caprylic acid fractionation. J Dairy Sci 96: 4535-4541.
- Abdou H, Marichatou H, Beckers JF, Hornick JL (2014) Effect of bovine colostrum administration on plasma protein profile, growth, and survival in red kid. Small Rumin Res 117: 158-164.
- Alvarez V, Arranz J, Daltabuit-Test M, Leginagoikoa I, Juste RA, et al. (2005) Relative contribution of colostrum from Maedi-Visna virus (MVV) infected ewes to MVV-seroprevalence in lambs. Res Vet Sci 78: 237-243.
- Konold T, Moore SJ, Bellworthy SJ, Terry LA, Thorne L, et al. (2013) Evidence of effective scrapie transmission via colostrum and milk in sheep. BMC Vet Res 9: 99.
- Hernández-Castellano LE, Suárez-Trujillo A, Martell-Jaizme D, Cugno G, Argüello A, et al. (2015) The effect of colostrum period management on BW and immune system in lambs: from birth to weaning. Animal 9: 1672-1679.
- Alves AC, Alves NG, Ascari IJ, Junqueira FB, Coutinho AS, et al. (2015) Colostrum composition of Santa Ines sheep and passive transfer of immunity to lambs. J Dairy Sci 98: 3706-3716.

Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences